CareTaker Medical's continuous blood pressure monitor gets 2nd FDA clearance

CareTaker Medical's continuous blood pressure monitor gets 2nd FDA clearance
The FDA granted a second 510(k) clearance this week to CareTaker, a Charlottesville, Virginia connected medical device company. CareTaker's device of the same name is a wearable, connected blood pressure and heart rate sensor that originally received clearance this time last year. “CareTaker is a real game changer, allowing physicians to remotely monitor medical-grade continuous blood pressure and heart rate from anywhere, using only a patient friendly-finger cuff,” Dr.

Caretaker Medical (@caretakermed) / X

Examples of FDA-approved devices: (a) the Visi mobile system (Sotera
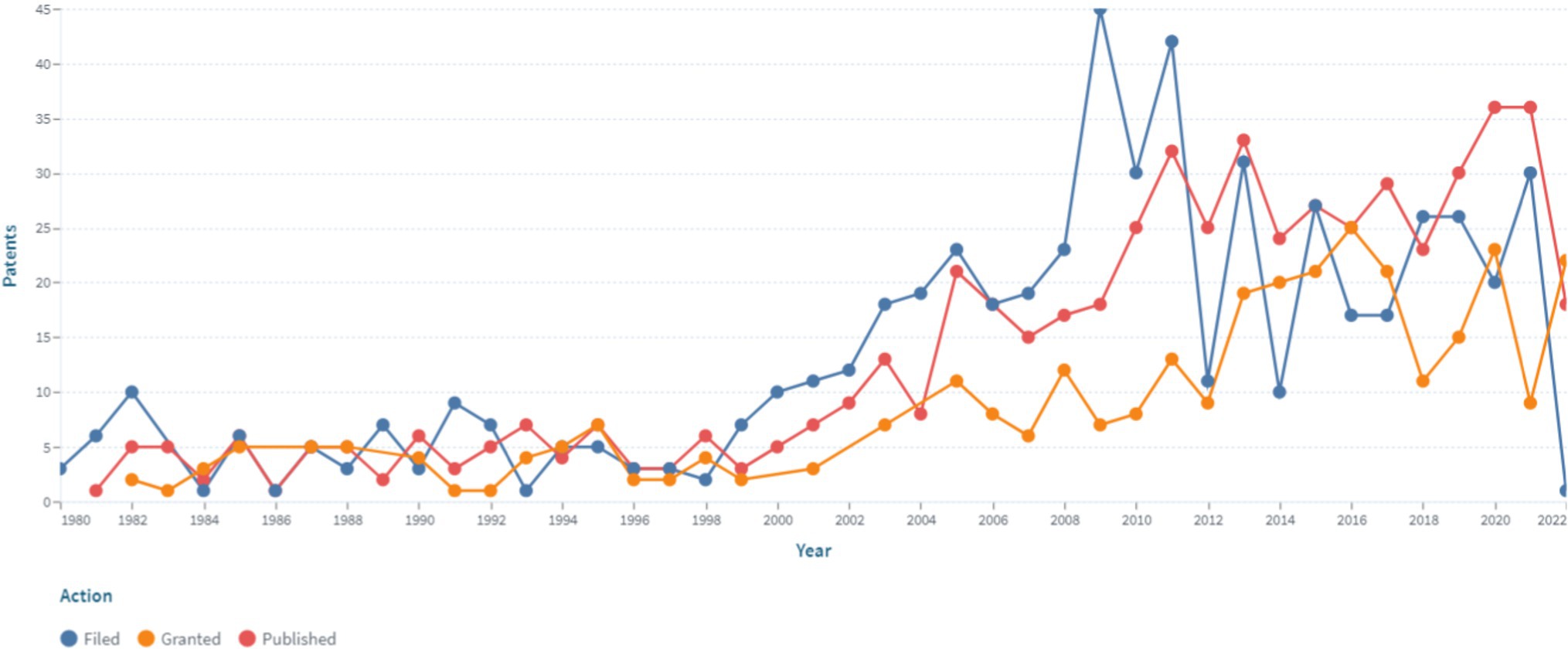
Frontiers Patent landscape review of non-invasive medical sensors for continuous monitoring of blood pressure and their validation in critical care practice

Caretaker Vitalsense Wrist Beat-by-Beat Blood Pressure Monitor, 0.01 ( pressure)
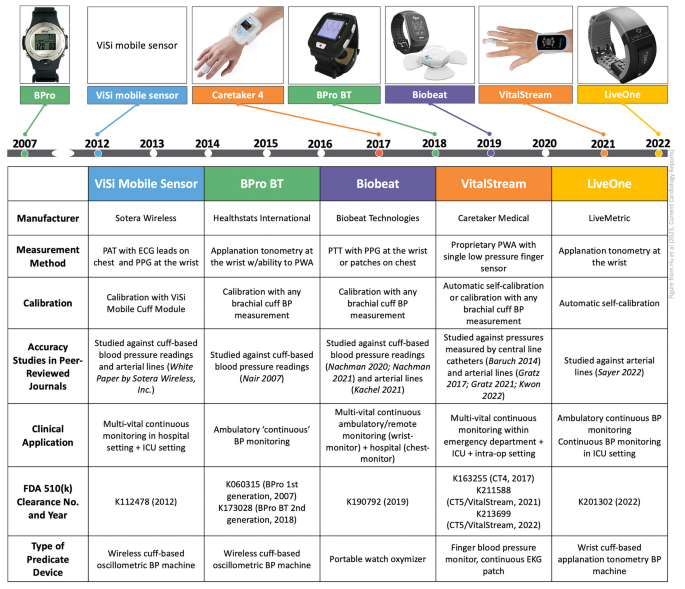
The Promise and Illusion of Continuous, Cuffless Blood Pressure Monitoring
A guide to connected health device and remote patient monitoring vendors

FDA Clears Caretaker Medical's Wireless Monitor for Continuous, Non-Invasive Cardiac Output, Stroke Volume, and Advanced Hemodynamics

Caretaker Medical Wins Certification for CNIBP
FDA Clearance for Capsule Monitor with Masimo Capnography

Downloads Caretaker Medical

![PDF] Continuous Non-Invasive Blood Pressure Monitoring: A Methodological Review on Measurement Techniques PDF] Continuous Non-Invasive Blood Pressure Monitoring: A Methodological Review on Measurement Techniques](https://d3i71xaburhd42.cloudfront.net/8e781b7ff3c43a92c8c5b0a02ae09ed1ec6e34be/2-Figure1-1.png)
