Side-chain hydrophobicity scale derived from transmembrane protein folding into lipid bilayers

Side-chain hydrophobicity scale derived from transmembrane protein folding into lipid bilayers
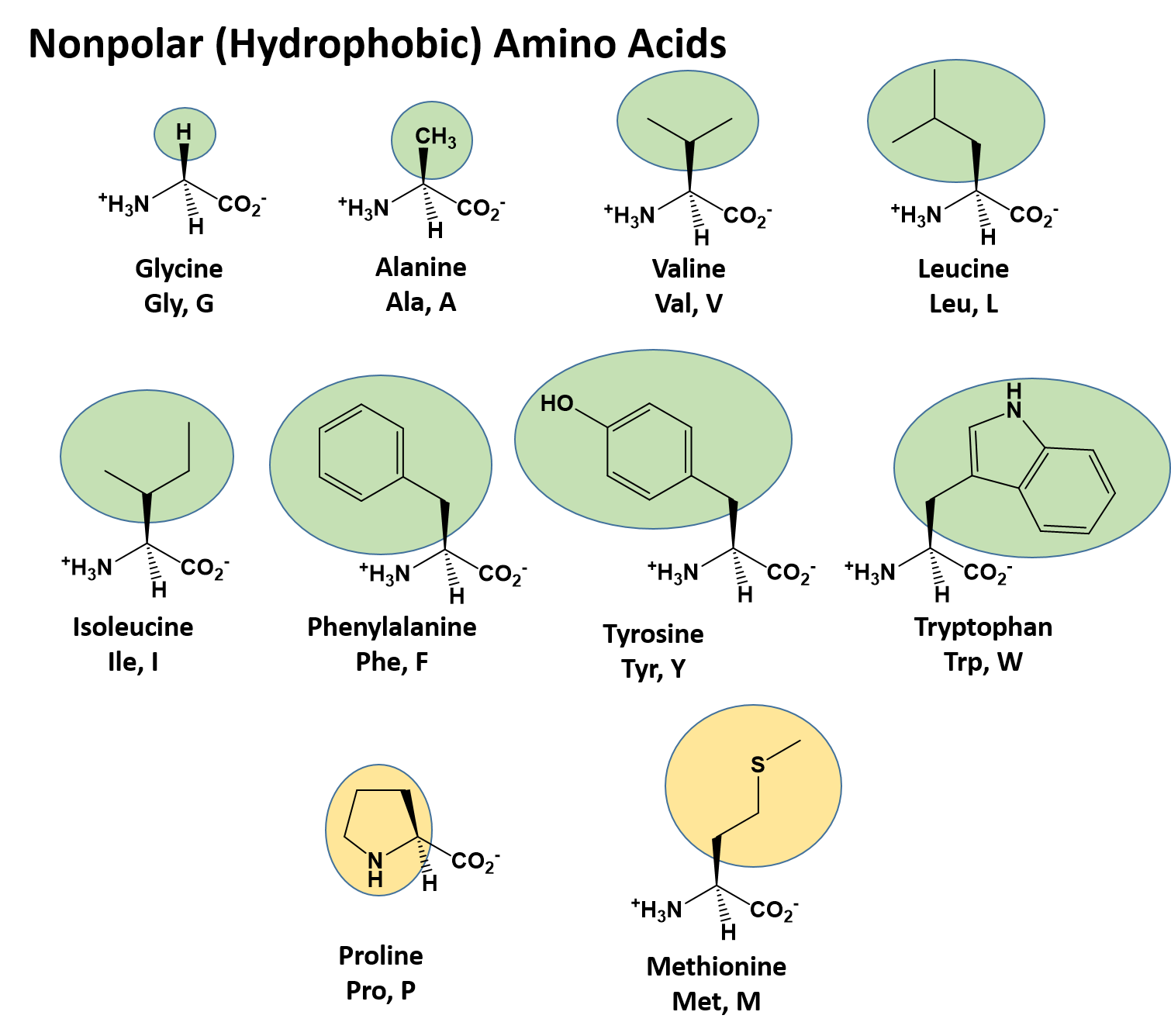
Chapter 2: Protein Structure - Chemistry

Chapter 11 Exam I Part 3 (C) Flashcards

The Hydrophobic Effect Is a Principal Force Stabilizing Tertiary and Quaternary Structures - LabXchange

Mechanisms of integral membrane protein insertion and folding. - Abstract - Europe PMC

Transmembrane protein - Wikipedia

Hydrophobicity Scales: Most Up-to-Date Encyclopedia, News & Reviews

Role of the lipid bilayer in outer membrane protein folding in Gram-negative bacteria - ScienceDirect
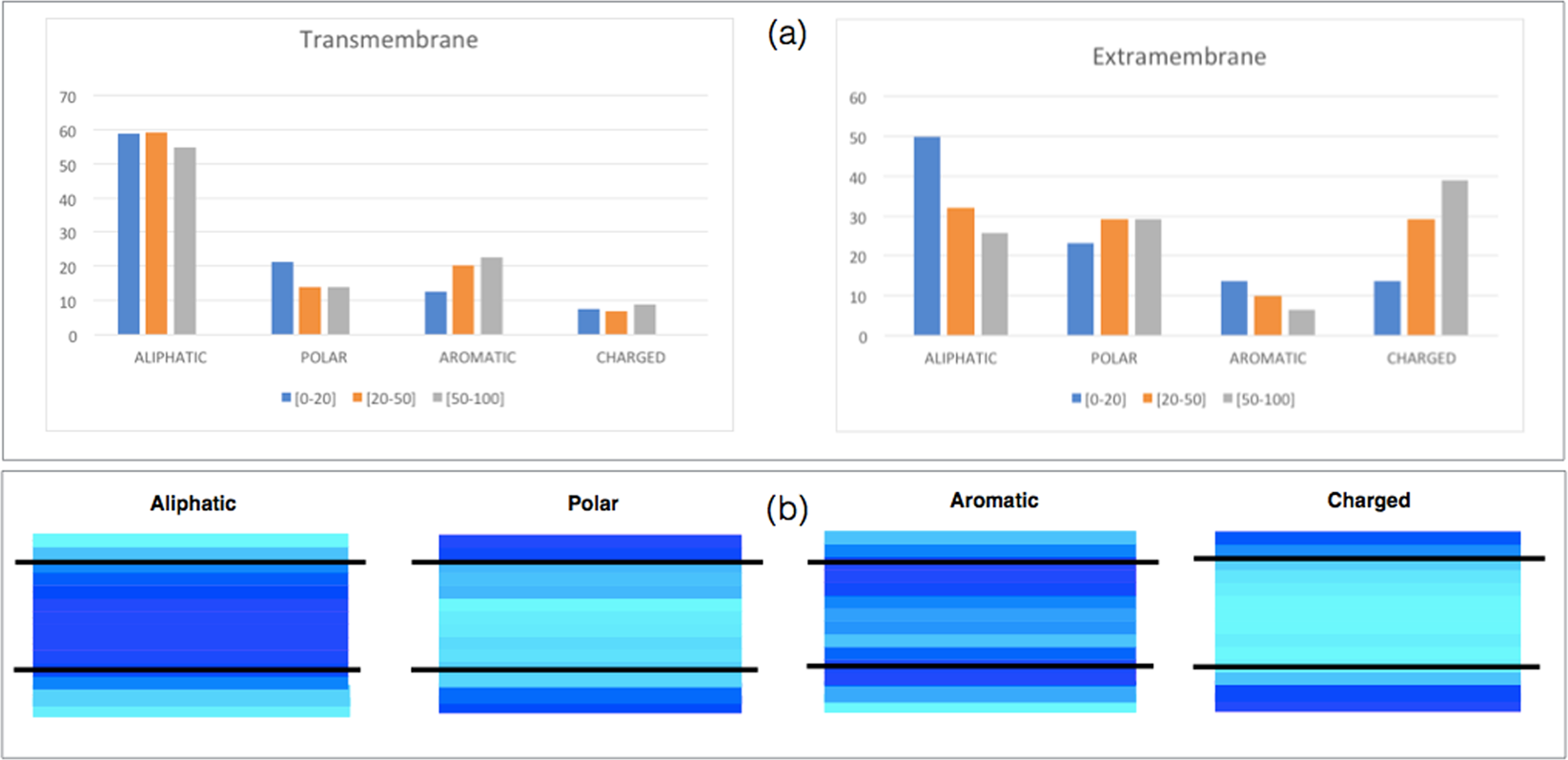
A comprehensive computational study of amino acid interactions in membrane proteins

6. Biological Membranes Contain Proteins as Well as Lipids - LabXchange
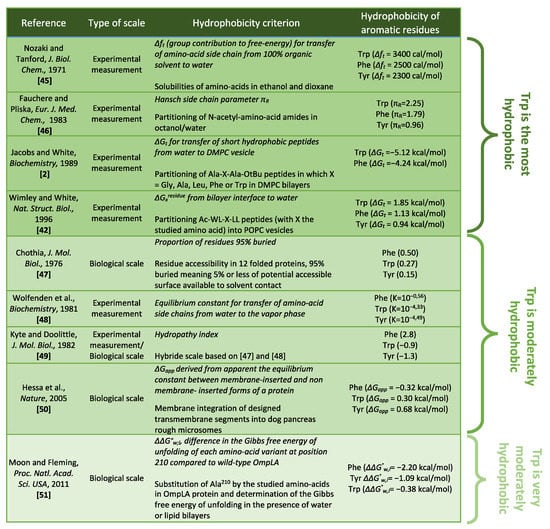
Crystals, Free Full-Text

Cell-Free Synthesis of a Transmembrane Mechanosensitive Channel Protein into a Hybrid-Supported Lipid Bilayer

Side-chain hydrophobicity scale derived from transmembrane protein folding into lipid bilayers
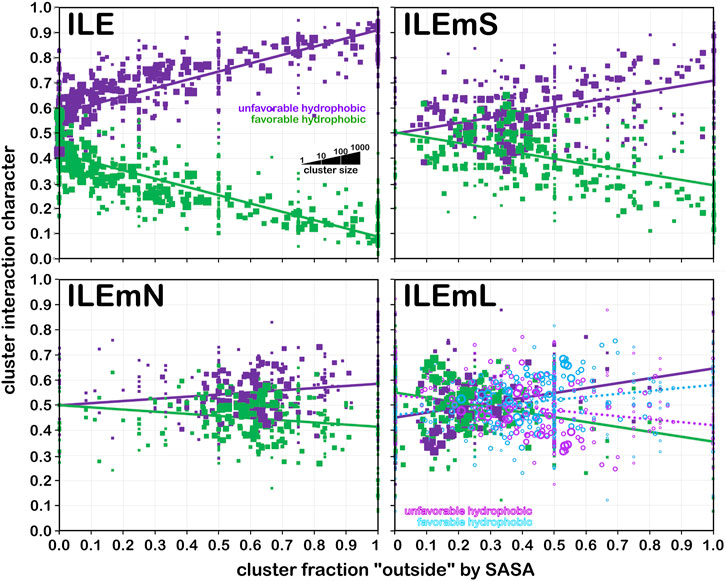
Frontiers 3D interaction homology: The hydrophobic residues alanine, isoleucine, leucine, proline and valine play different structural roles in soluble and membrane proteins

Marginally hydrophobic transmembrane α‐helices shaping membrane protein folding - De Marothy - 2015 - Protein Science - Wiley Online Library

IJMS, Free Full-Text


