FDA clears Sotera's continuous blood pressure monitor

FDA clears Sotera's continuous blood pressure monitor
Sotera Wireless has received yet another FDA 510(k) clearance for its ViSi remote patient monitoring system, this time for its novel continuous noninvasive blood pressure (cNIBP) monitoring software. The technology, which enables continuous blood pressure monitoring without a cuff or catheter, has been part of Sotera's pitch from the beginning, but was not included in the company's April 2012 device clearance or its August 2012 full system clearance.

Sotera Wireless Seeks Continuous Vital Sign Monitoring
Features, 1.Fully automatic blood pressure measure, 2.The Electronic Sphygmomanometer stores the measure results of three users automatically, and up
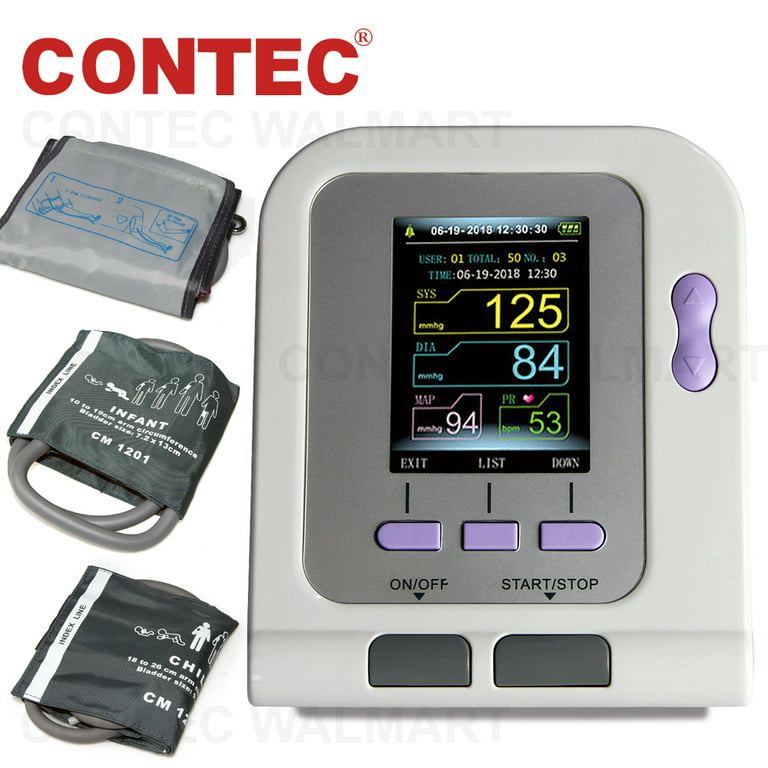
FDA Approved Fully Automatic Upper Arm Blood Pressure Monitor 3 mode 3 cuffs Electronic Sphygmomanometer

Examples of FDA-approved devices: (a) the Visi mobile system
FDA clears LiveMetric's smartwatch-like blood pressure sensor

Validating cuffless continuous blood pressure monitoring devices

Aktiia Blood Pressure Monitor Launches in U.S.

The Microsoft Research Aurora Project: Important Findings on
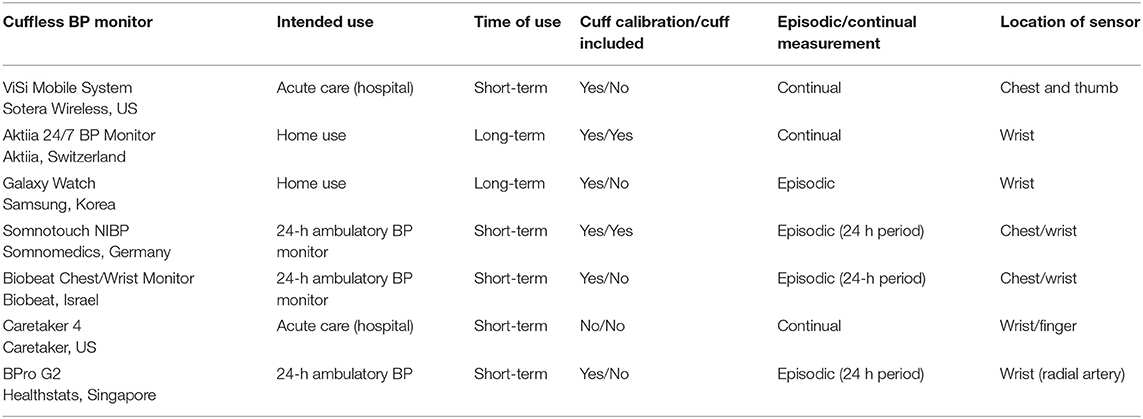
Frontiers Guidance for the Interpretation of Continual Cuffless
Features, 1.Fully automatic blood pressure measure, 2.The Electronic Sphygmomanometer stores the measure results of three users automatically, and up
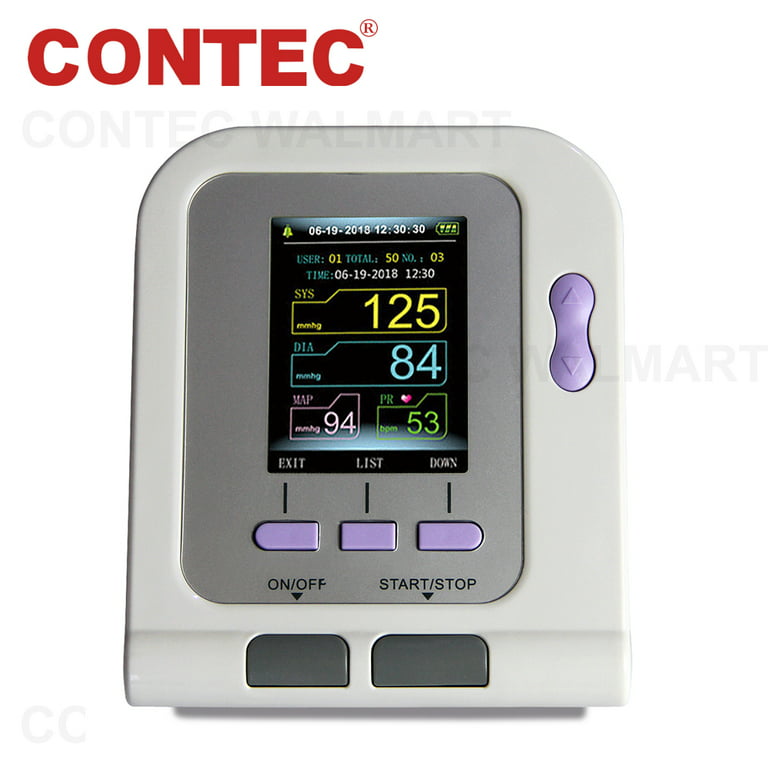
FDA Approved Fully Automatic Upper Arm Blood Pressure Monitor 3 mode 3 cuffs Electronic Sphygmomanometer
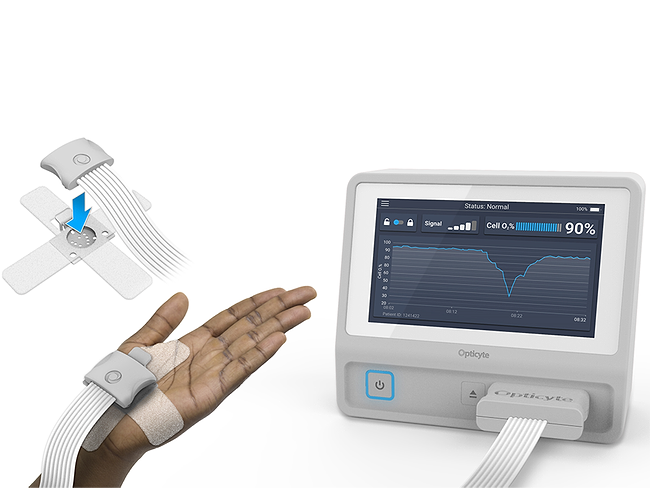
Patient monitoring
Sotera Digital Health Launches ViSi Mobile for Early Detection


