Tackling Salami Slicing and Indication Stacking in Orphan Drug Innovation Incentives - Bill of Health

Tackling Salami Slicing and Indication Stacking in Orphan Drug Innovation Incentives - Bill of Health
Are the incentive mechanisms for orphan drug development working as they should?

Tackling Salami Slicing and Indication Stacking in Orphan Drug Innovation Incentives - Bill of Health

Orphan Drugs in Oncology

Drugmakers Manipulate Orphan Drug Rules To Create Prized Monopolies - KFF Health News

Health Justice as the Lodestar of Incremental Health Reform - Bill of Health
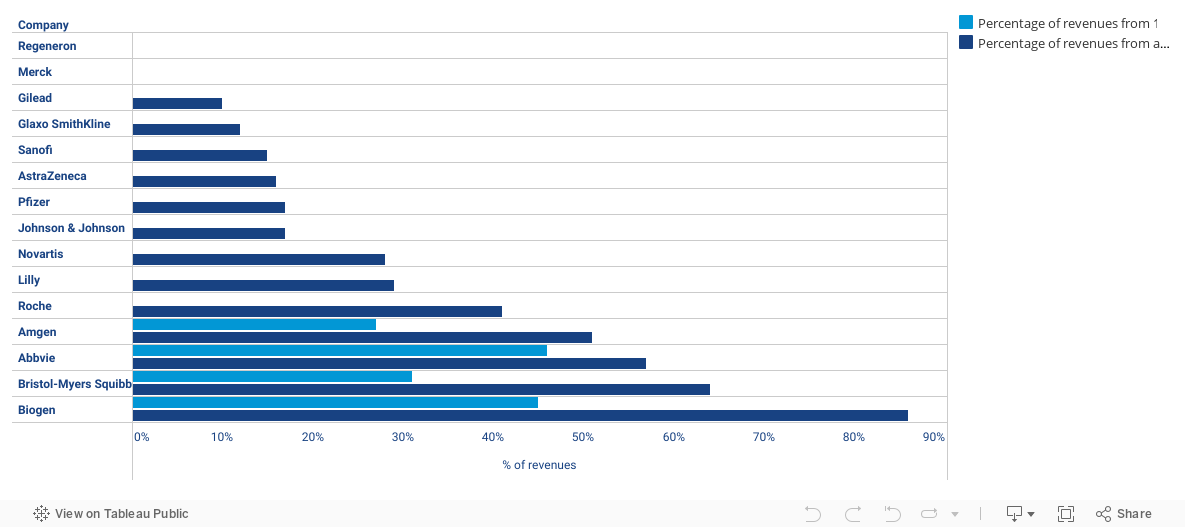
Abuses of Government Programs and Regulations - Drug Pricing Lab

Incentives - Bill of Health

How Orphan Drugs Became a Highly Profitable Industry

How Orphan Drugs Became a Highly Profitable Industry

Salami-slicing, precision medicine and the Orphan Drug Act

Salami-slicing, precision medicine and the Orphan Drug Act

ICER Publishes White Paper Evaluating Reforms to Orphan Drug Development, Pricing, and Coverage - ICER
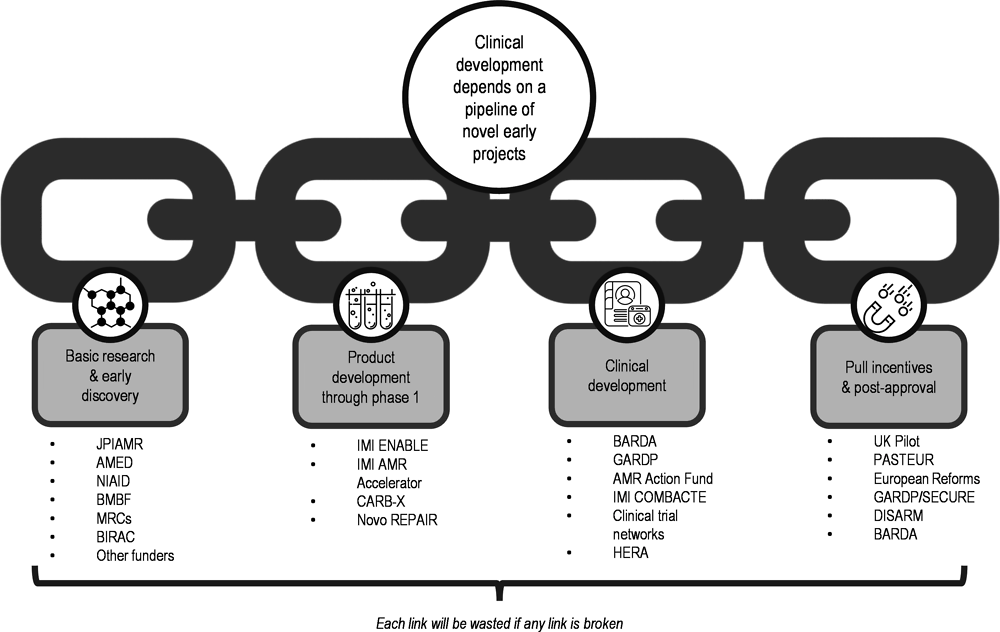
13. Incentivising the Development of Global Public Goods for Health, Ready for the Next Crisis? Investing in Health System Resilience

Frontiers Orphan Medicine Incentives: How to Address the Unmet Needs of Rare Disease Patients by Optimizing the European Orphan Medicinal Product Landscape Guiding Principles and Policy Proposals by the European Expert

FDA approval, clinical trial evidence, efficacy, epidemiology, and price for non-orphan and ultra-rare, rare, and common orphan cancer drug indications: cross sectional analysis
GHB, Orphans, and REMS: A Regulatory Love Story (Part 1)


