Ice - Structure, Formation, Properties

Ice - Structure, Formation, Properties
Ice - Structure, Formation, Properties: At standard atmospheric pressure and at temperatures near 0 °C, the ice crystal commonly takes the form of sheets or planes of oxygen atoms joined in a series of open hexagonal rings. The axis parallel to the hexagonal rings is termed the c-axis and coincides with the optical axis of the crystal structure. When viewed perpendicular to the c-axis, the planes appear slightly dimpled. The planes are stacked in a laminar structure that occasionally deforms by gliding, like a deck of cards. When this gliding deformation occurs, the bonds between the layers break, and the hydrogen atoms involved in those.
Ice, solid substance produced by the freezing of water vapour or liquid water. At temperatures below 0 °C (32 °F), water vapour develops into frost at ground level and snowflakes (each of which consists of a single ice crystal) in clouds. Below the same temperature, liquid water forms a solid, as

Formation of hexagonal and cubic ice during low-temperature growth
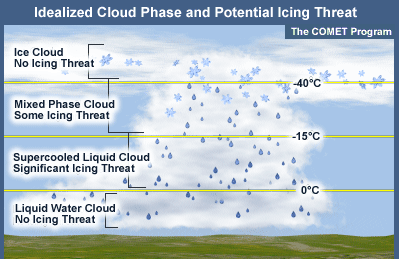
Icing Hazards

Antarctic Landfast Sea Ice: A Review of Its Physics, Biogeochemistry and Ecology - Fraser - 2023 - Reviews of Geophysics - Wiley Online Library

Describing the Properties of Ionic Solids, Chemistry
Structure of Water and Hydrogen Bonding
Lesson 10: Properties of Water

The Crystalline Structure of Ice, Journal of Glaciology

Why Does Water Expand When It Freezes? » Science ABC

Compound Interest: The Chemistry of Ice Cream – Components, Structure, & Flavour

What is a Crystal? Let's Talk Science

14.1 Types of Ice – Introduction to Oceanography

Medium-density amorphous ice
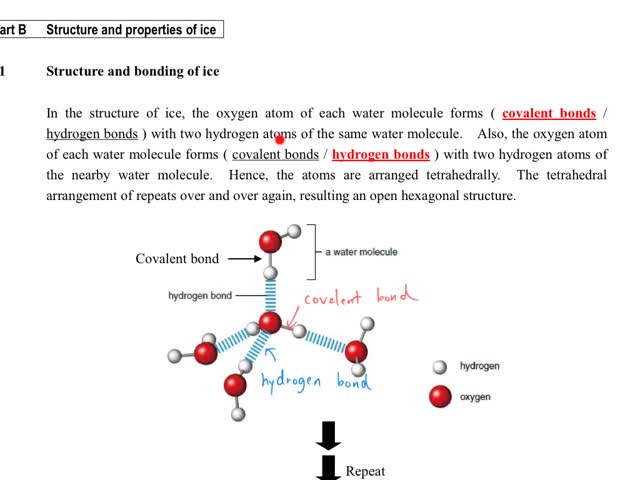
Chemistry tutorial-Ch27-2-Structure and bonding of ice

