FDA gives nod to first fully-removable percutaneous peripheral

FDA gives nod to first fully-removable percutaneous peripheral
SPR Therapeutics has developed a peripheral nerve stimulation system that is placed percutaneously through the skin instead of being implanted and which can be completely removed from the body after therapy period.
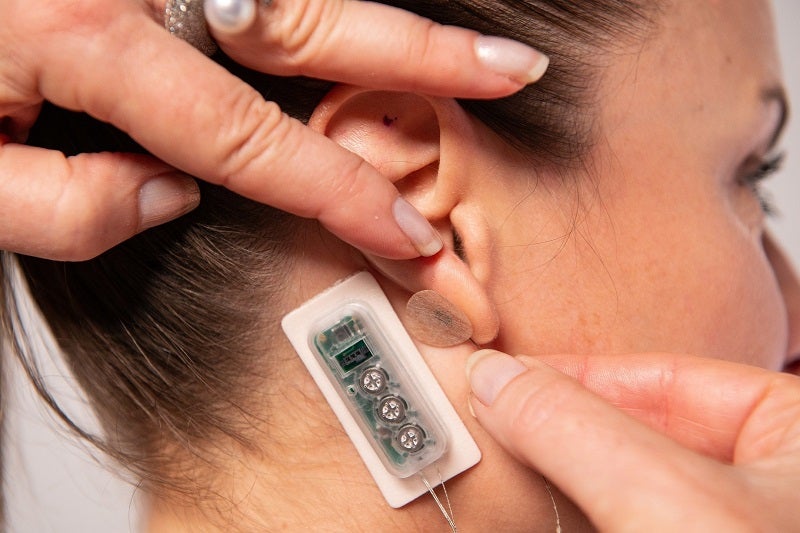
DyAnsys receives US FDA approval for First Relief neurostimulation device
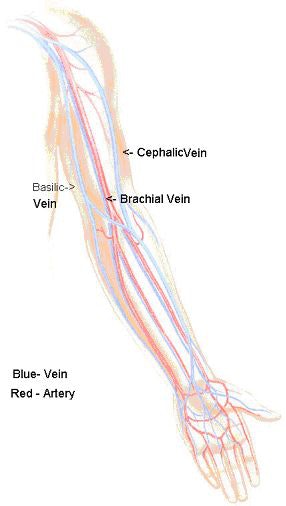
PICCing sides: Interventional radiologists weigh IV access lines

Locoregional delivery of CAR-T cells in the clinic - ScienceDirect
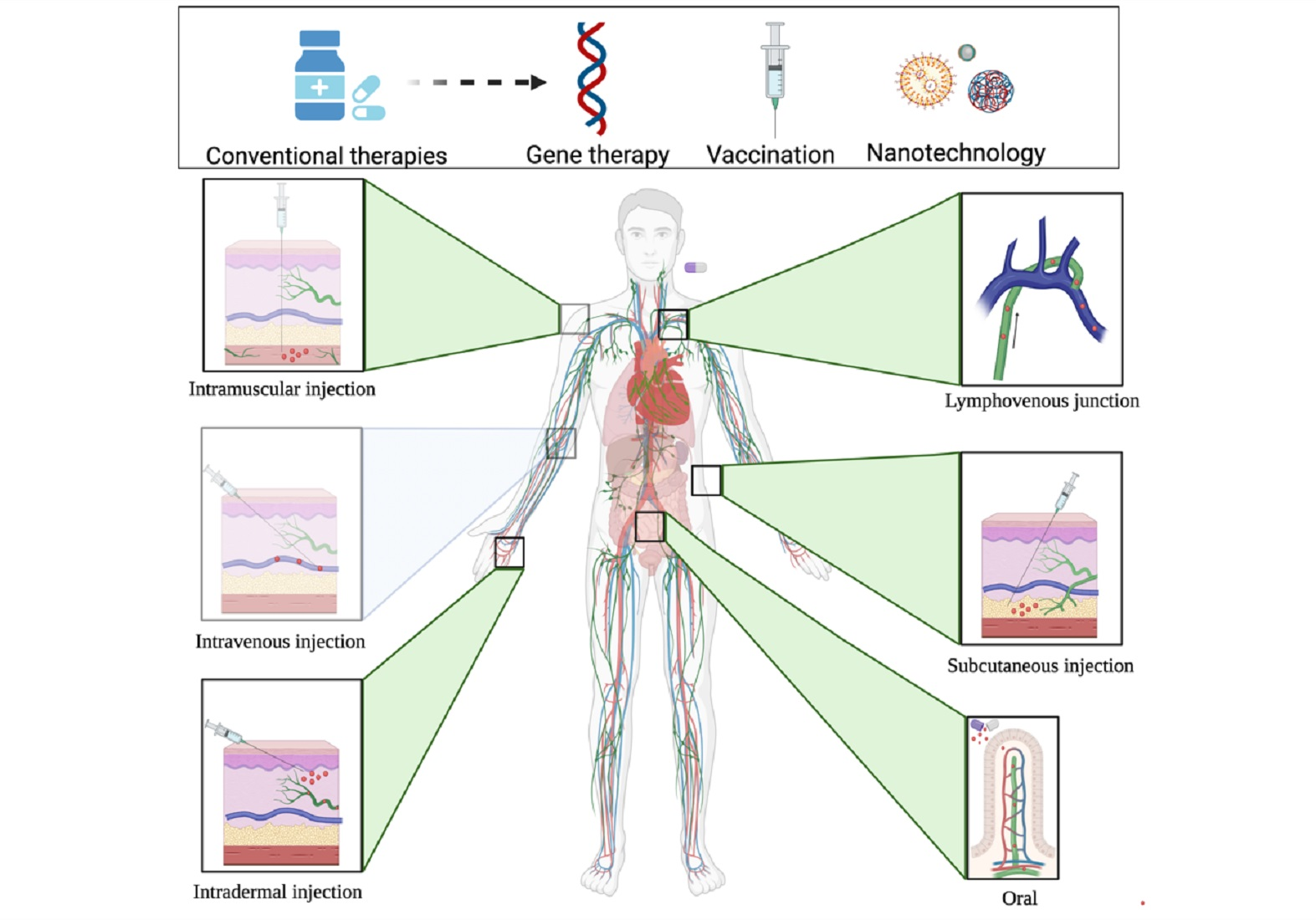
Pharmaceutics, Free Full-Text

Microparticles: biogenesis, characteristics and intervention therapy for cancers in preclinical and clinical research, Journal of Nanobiotechnology

urmila, Author at Interventional News

Smart Contact Lenses as Wearable Ophthalmic Devices for Disease Monitoring and Health Management
VOMIT APE PR;;;;@ - Market Show of Best Club crypto NFT ; Ape ; ARt ; gif

NDI Medical - Crunchbase Company Profile & Funding


