Cuffless Blood Pressure Monitors: Principles, Standards and Approval for Medical Use

Cuffless Blood Pressure Monitors: Principles, Standards and Approval for Medical Use
The principles of cuffless BP monitors are described, and the current situation regarding BP monitor standards and approval for medical use is discussed. SUMMARY Cuffless blood pressure (BP) monitors are noninvasive devices that measure systolic and diastolic BP without an inflatable cuff. They are easy to use, safe, and relatively accurate for resting-state BP measurement. Although commercially available from online retailers, BP monitors must be approved or certificated by medical regulatory bodies for clinical use. Cuffless BP monitoring devices also need to be approved; however, only the Institute of Electrical and Electronics Engineers (IEEE) certify these devices. In this paper, the principles of cuffless BP monitors are described, and the current situation regarding BP monitor standards and approval for medical use is discussed.

Long-term stability of over-the-counter cuffless blood pressure monitors: a proposal
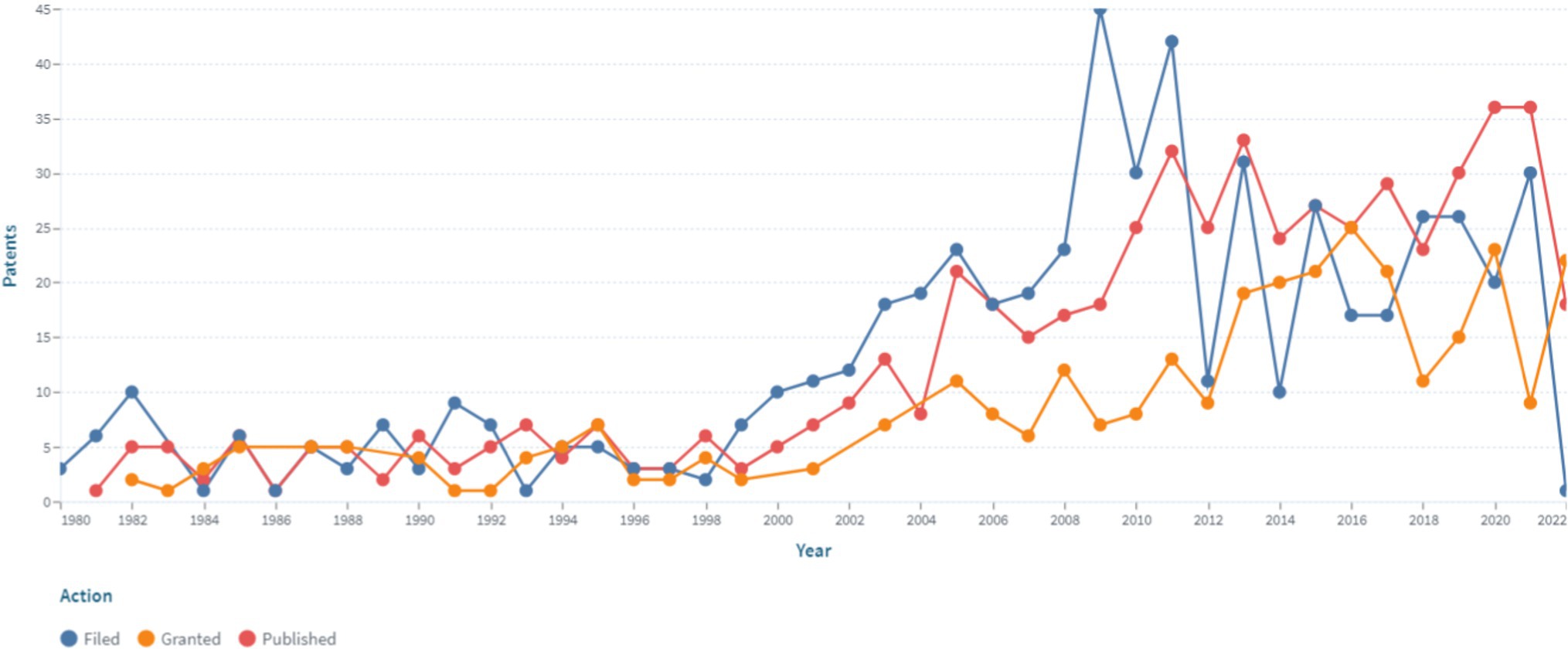
Frontiers Patent landscape review of non-invasive medical sensors for continuous monitoring of blood pressure and their validation in critical care practice

Continuous non-invasive monitoring of blood pressure in the operating room: a cuffless optical technology at the fingertip
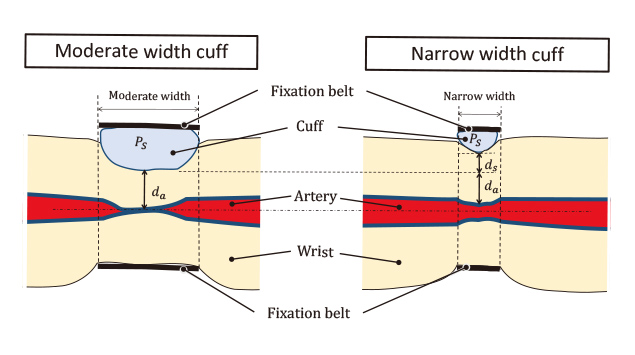
Arterial Compression Technology for a Watch-type Blood Pressure Monitor, OMRON TECHNICS, Technology

Validating cuffless continuous blood pressure monitoring devices - Cardiovascular Digital Health Journal

The Microsoft Research Aurora Project: Important Findings on Cuffless Blood Pressure Measurement

Full article: Ambulatory blood pressure monitoring by a novel cuffless device: a pilot study

Validating cuffless continuous blood pressure monitoring devices - ScienceDirect

Blood pressure - Wikipedia

Cuffless Blood Pressure Monitors: Approved but Unproven

PDF) Validating cuffless continuous blood pressure monitoring devices