Senza Spinal Cord Stimulation System – P130022/S039
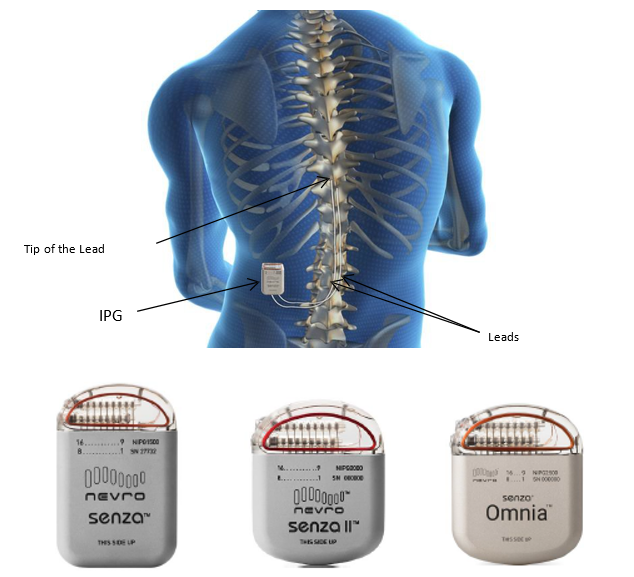
Senza Spinal Cord Stimulation System – P130022/S039
The Senza, Senza II, and Senza Omnia are implanted, rechargeable Spinal Cord Stimulation systems to treat chronic pain in a patient’s trunk or limbs that is difficult to manage.
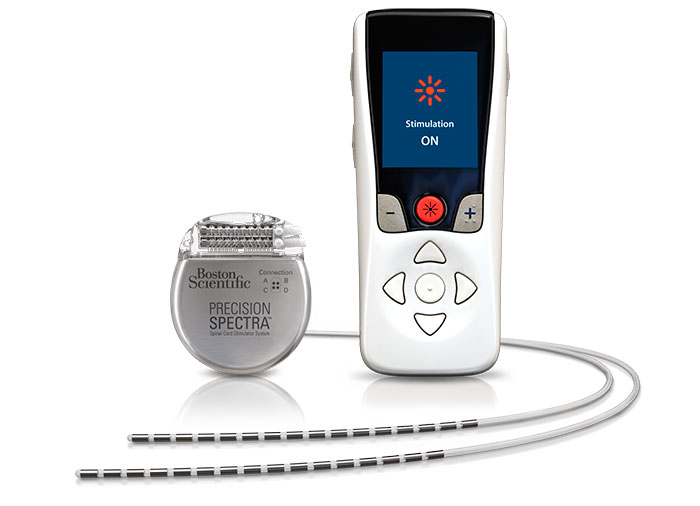
Spinal Cord Stimulation Trial

Nevro Senza - Omnia - Spinal Cord Stimulator (SCS) System By

Spinal Cord Stimulation – The World of Implantable Devices

FDA approves Nevro spinal cord stimulation to treat chronic pain with diabetic neuropathy - Drug Delivery Business

Nevro receives CE mark for full-body MRI conditional labelling with the Senza system - NeuroNews International

FDA approves new leads for Nevro's Senza spinal cord stimulator - MassDevice

Nevro Receives FDA Approval For Senza II Spinal Cord Stimulation System Delivering HF10 Therapy

FDA Approves Spinal Cord Stimulation Therapy for Painful Diabetic Neuropathy

Spinal cord stimulation
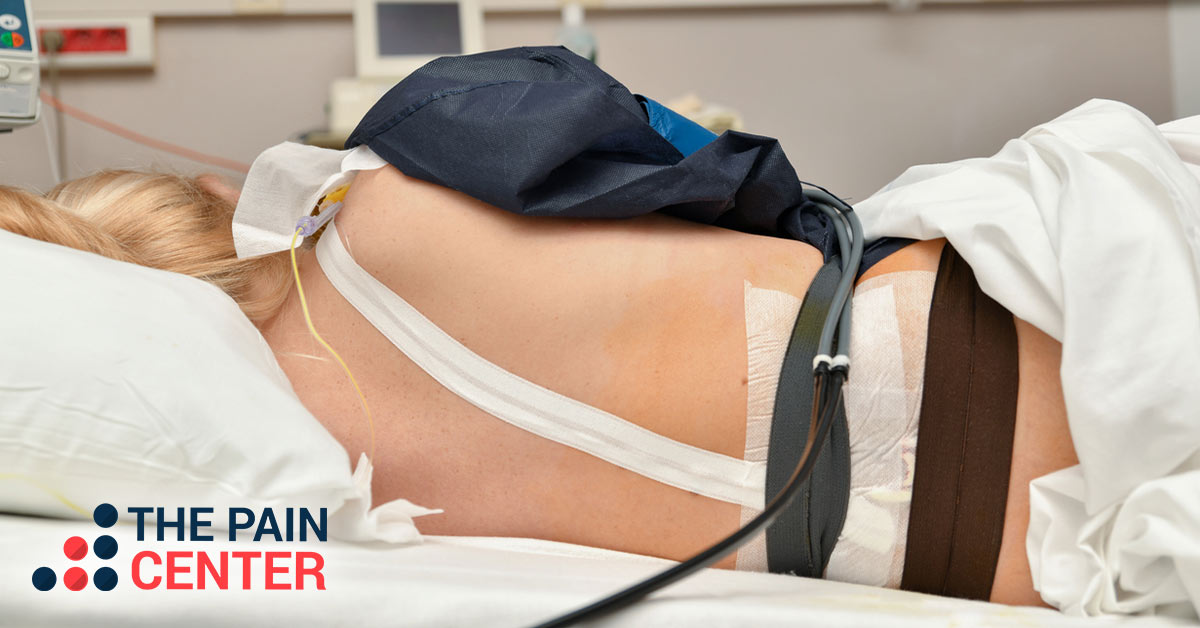
Spinal Cord Simulator, Chronic Back Pain

Nevro Corp. - US - Providers - Product: Trial Stimulator

Spinal-cord stimulators help some patients, injure others

St. Jude Medical™ Nicht sichtbares Testsystem für SCS

What Is Neuropathy? Symptoms, Causes, Diagnosis, Treatment And Prevention – Forbes Health
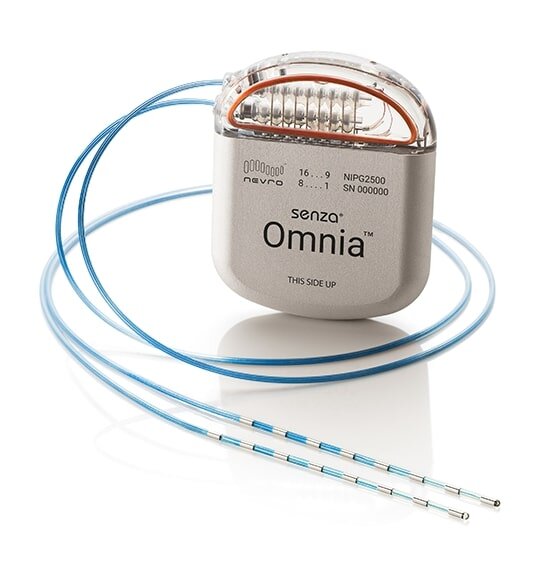
FDA Approves Spinal Cord Stimulator for Diabetic Neuropathy — Pain News Network


/assets/images/provider/photos/2600332.jpg)