Cuffless Blood Pressure Monitors: Principles, Standards and

Cuffless Blood Pressure Monitors: Principles, Standards and
The principles of cuffless BP monitors are described, and the current situation regarding BP monitor standards and approval for medical use is discussed. SUMMARY Cuffless blood pressure (BP) monitors are noninvasive devices that measure systolic and diastolic BP without an inflatable cuff. They are easy to use, safe, and relatively accurate for resting-state BP measurement. Although commercially available from online retailers, BP monitors must be approved or certificated by medical regulatory bodies for clinical use. Cuffless BP monitoring devices also need to be approved; however, only the Institute of Electrical and Electronics Engineers (IEEE) certify these devices. In this paper, the principles of cuffless BP monitors are described, and the current situation regarding BP monitor standards and approval for medical use is discussed.
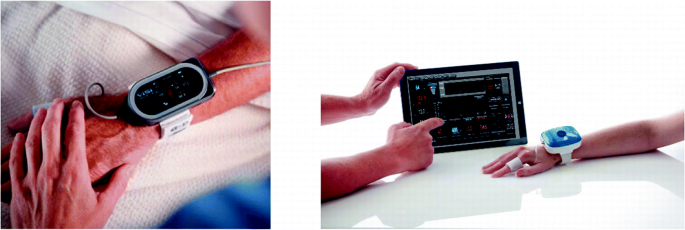
Regulation and Approval of Continuous Non-invasive Blood-Pressure Monitoring Devices
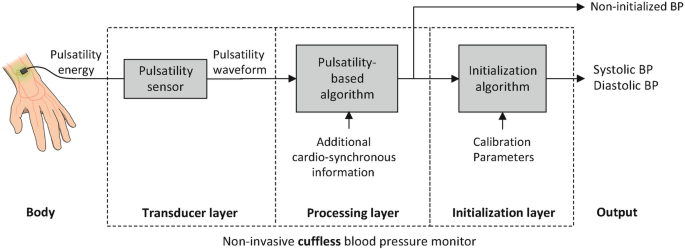
The Definition and Architecture of Cuffless Blood Pressure Monitors

Figure 2 from SeismoWatch
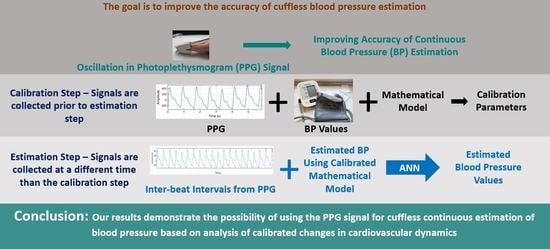
Bioengineering, Free Full-Text
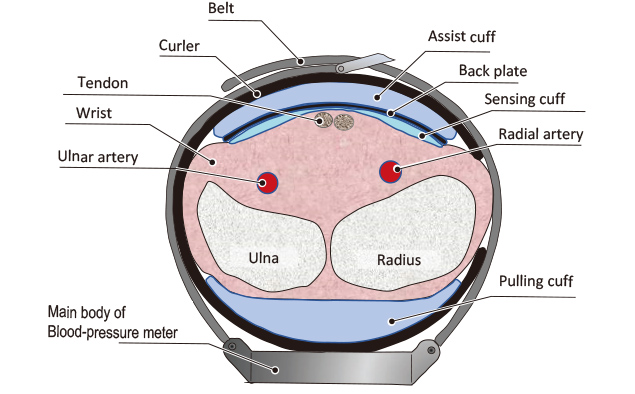
Arterial Compression Technology for a Watch-type Blood Pressure Monitor, OMRON TECHNICS, Technology
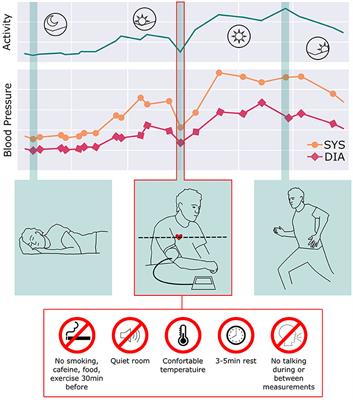
Frontiers Guidance for the Interpretation of Continual Cuffless Blood Pressure Data for the Diagnosis and Management of Hypertension

Japanese Medical Device Nomenclature (JMDN) newly introduced on July
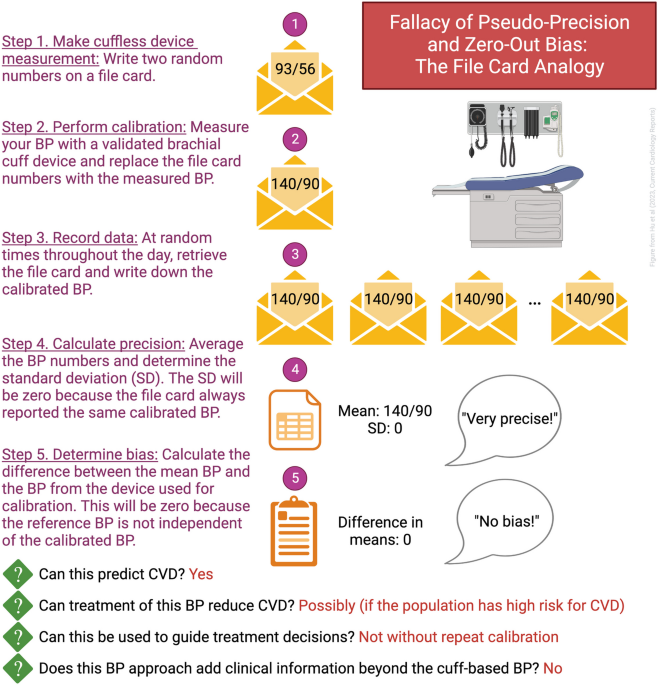
The Promise and Illusion of Continuous, Cuffless Blood Pressure Monitoring

Cuff-Less Methods for Blood Pressure Telemonitoring

Continuous non-invasive monitoring of blood pressure in the operating room: a cuffless optical technology at the fingertip
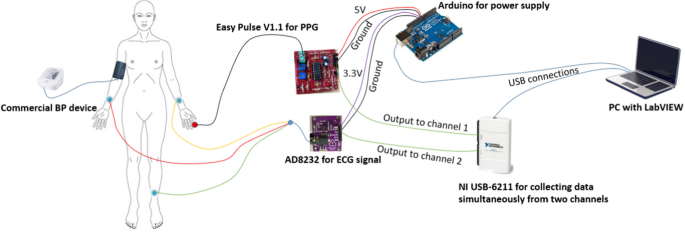
Non-invasive cuff-less blood pressure estimation using a hybrid deep learning model

Swiss startup Aktiia scores over $6M for its cuff-less blood monitoring system
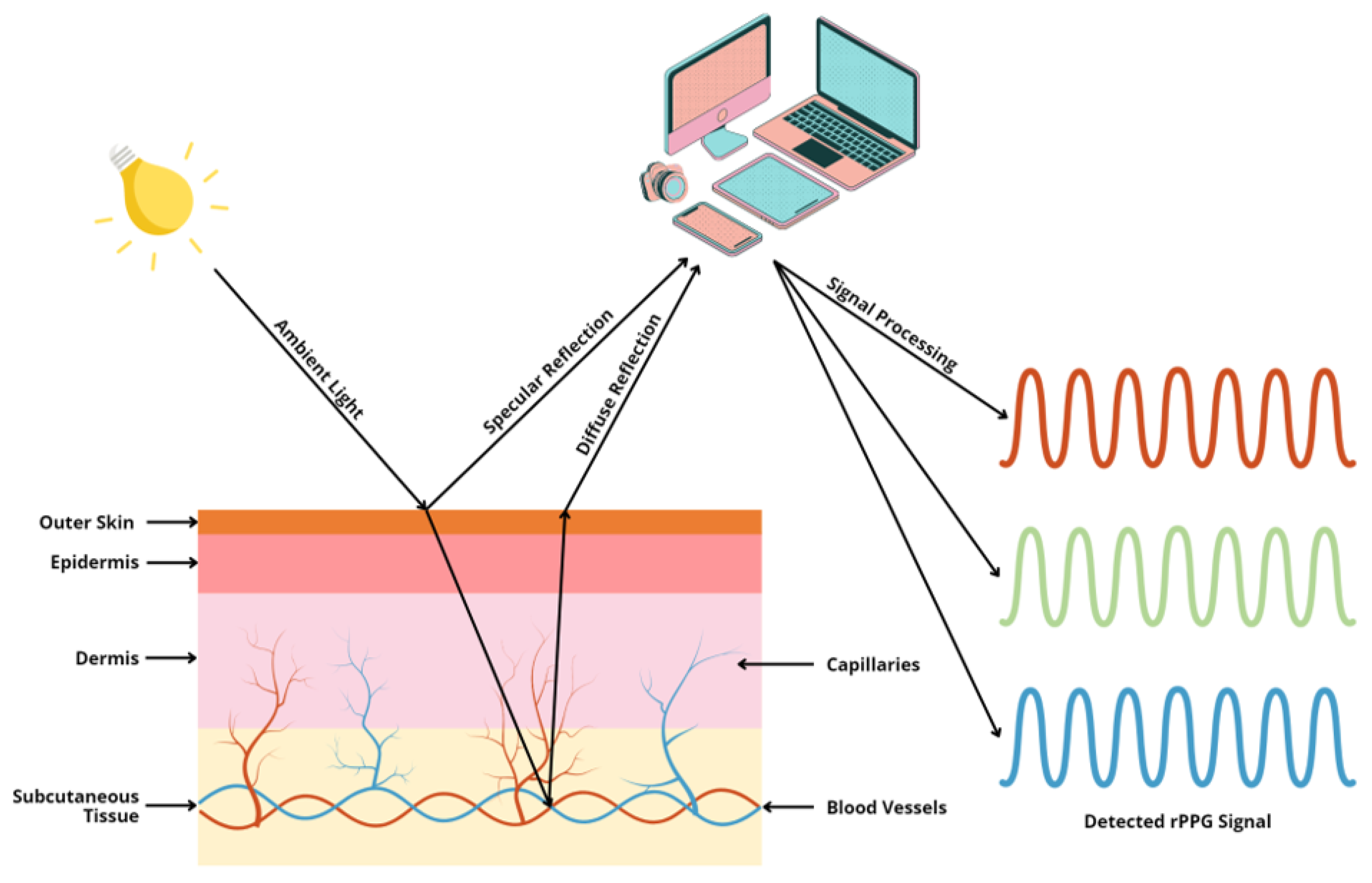
Healthcare, Free Full-Text